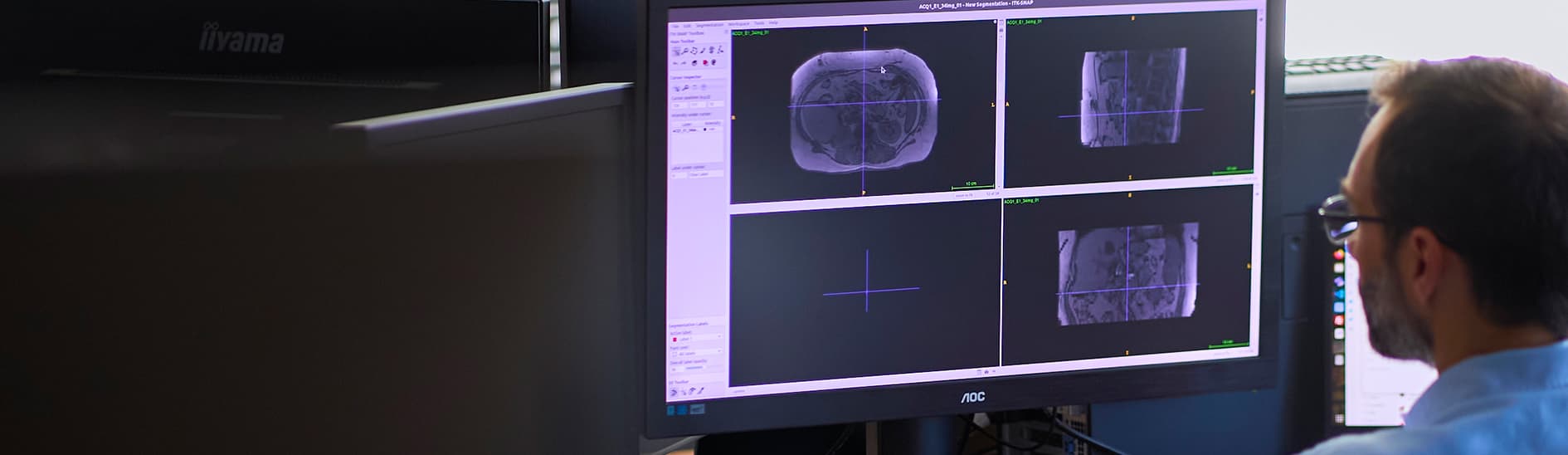
Liver fibrosis
NAFLD/NASH
NAFLD encompasses the spectrum of liver disease, ranging from non-alcoholic fatty liver (NAFL) to non-alcoholic steatohepatitis (NASH), fibrosis and cirrhosis
Patients at risk of NAFLD and NASH are not always identified as there are no obvious clinical manifestations and it can remain silent for several years as it progresses
Fibrosis is becoming the main predictor of NAFLD progression to NASH and if detected at an early stage it can be prevented through lifestyle changes
Currently, there is no single alternative that can provide all the information offered by a liver biopsy – consequently, there is an urgent need for non-invasive and cost-effective biomarkers to facilitate widespread surveillance
Median’s eyonis™ NASH
Unrivalled AI/ML Software as Medical Device (SaMD) enabling liver fibrosis diagnosis with unprecedented performance
The only end-to-end AI/ML tech based SaMDs indicated for liver fibrosis in NASH
Unprecedented sensitivity and specificity performance
eyonis™ has the potential to identify fibrosis when still reversible
Additional indications
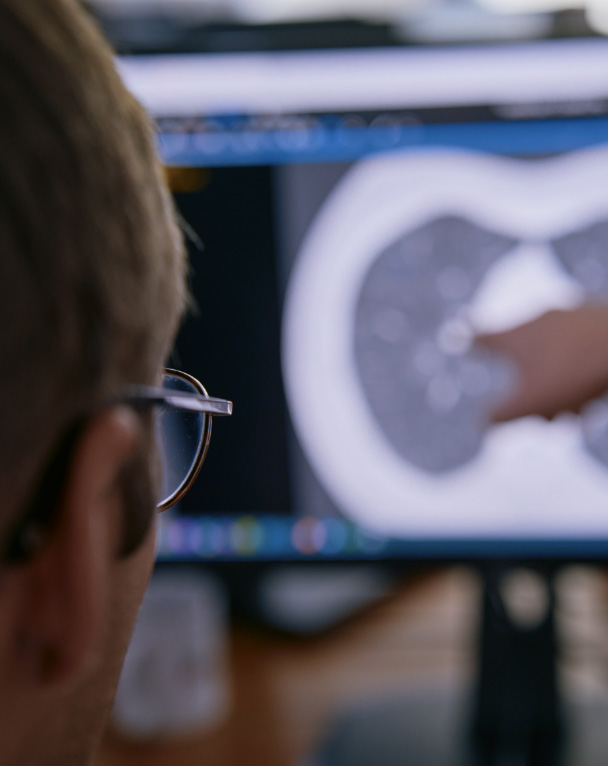
Lung cancer
Lung cancer screening (LCS) & incidental pulmonary nodules (IPN)

Liver cancer
Very early Hepatocellular Carcinoma (HCC)
Latest news

PRESS RELEASE
Median Technologies Announces Rebranding of iBiopsy®, Changes Name to eyonis™
Median Technologies unveils a new identity, including a new name, logo, tagline for its iBiopsy® AI/ML tech-based suite of software as medical devices (SaMD) and business unit, as part of an extensive rebranding initiative. iBiopsy® becomes eyonis™.

PRESS RELEASE
Median Technologies to speak at the Radiological Society of North America 2023 Annual Meeting, Nov. 26 – 30, Chicago, IL, USA
AI Theater Presentation: “Transforming Cancer Diagnosis through End-to-End AI-Powered SaMD”

PRESS RELEASE
First results of Median Technologies’ iBiopsy® HCC detection AI model developed on the PHELICAR Clinical Data Registry, to be presented at the ESMO Congress(Oct 20-24, 2023, Madrid, Spain)
