
Catching the unseen
Supporting radiologists and clinicians worldwide in screening, early diagnosis & treatment of cancer
Next generation end-to-end AI/ML tech-based CADe CADx Software as Medical Devices (SaMDs) with unprecedented performance
Our goal is to reduce cancers burdens by helping radiologists and clinicians identify disease at the earliest possible stage, allowing better care while avoiding unnecessary procedures and reducing healthcare costs.
With eyonis®, we are committed to improving patients’ lives with a portfolio of the most accurate AI/ML tech-based Software as Medical Devices (SaMDs) dedicated to early diagnosis of cancers.
A radiological CADe device is “intended to identify, mark, highlight or otherwise direct attention to portions of an image that may reveal abnormalities during interpretation of images by the clinician.” A CADx device is “intended to provide information beyond identifying abnormalities, such as an assessment of disease.” Source: FDA
eyonis®
Enabling the early detection & characterization of cancer to improve patients’ lives
Patient Benefits
Early and accurate diagnosis
Shorter time to treatment
Reduced disease burden and improved examination adherence
Clinician Benefits
Decrease false positives and false negative results
Reduced image review time and intra & inter-operator variability
Increased patient compliance thanks to greater trust in diagnostic accuracy
Healthcare Organizations Benefits
Fewer procedures to reach final diagnosis
Reduced need for late-stage & costly drug treatments
Hospitalizations reduction
Leveraging Median’s medical imaging expertise
Combining the power of AI & Data Science
eyonis® provides a portfolio of innovative end-to-end AI/ML tech-based Software as Medical Devices (SaMDs) in indications where early diagnosis is an unmet need.
eyonis®’s goal is to shift the medical diagnostic paradigm, in Lung Cancer and Liver Cancer.

eyonis® Workflow
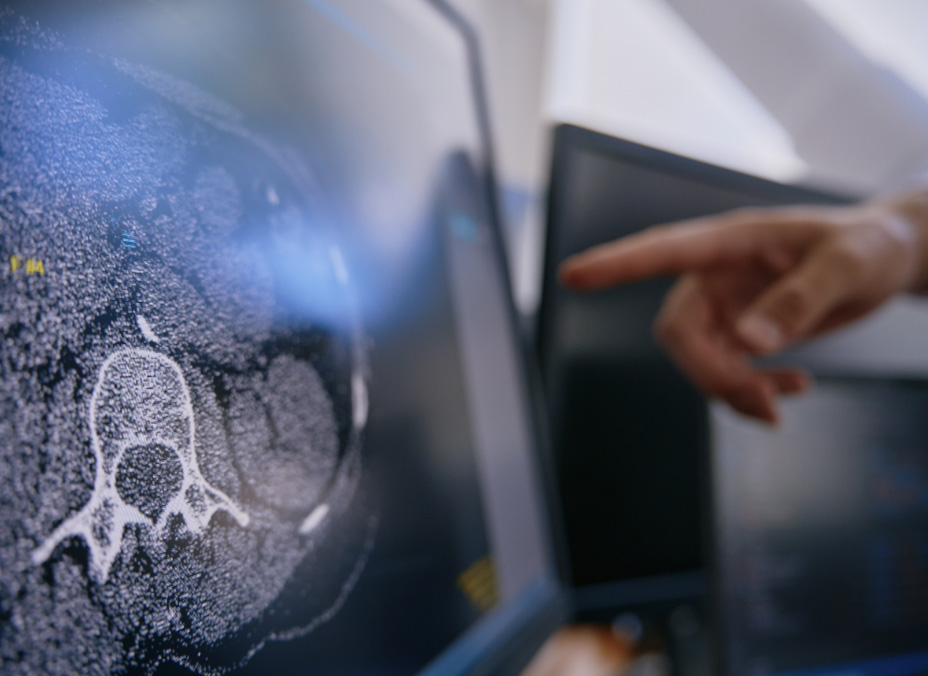
The power of eyonis®
Leveraging Median’s medical imaging expertise, combined with the power of AI & Data Science, eyonis® brings the power to:
- Achieve unprecedented diagnostic accuracy
- Decrease false negative & false positive results
- Improve patients’ lives
- Optimize radiologist time
- Increase workflow standardization
- Reduce unnecessary procedures and healthcare spending
eyonis® indications
Lung cancer
Lung Cancer Screening (LCS) and incidental pulmonary nodules (IPN)

Liver cancer
Very early Hepatocellular Carcinoma (HCC)
Latest news

PRESS RELEASE
Median Technologies achieves ISO 13485:2016 certification for eyonis® Medical Device Quality Management System

PRESS RELEASE
FDA Clearance Sets Stage for U.S. Commercialization of eyonis® LCS Software as a Medical Device for Lung Cancer Screening; Oran Muduroglu Appointed President of Median eyonis Inc.

PRESS RELEASE
Median Technologies announces collaboration with Tempus to expand access to eyonis® LCS Software as a Medical Device in the United States

PRESS RELEASE
Median Technologies receives FDA 510(k) clearance for eyonis® LCS, the first AI tech-based detection and diagnosis device for lung cancer screening

Learn more about eyonis®
Our team welcomes you to explore further our unprecedented technology for diagnostic imaging.
